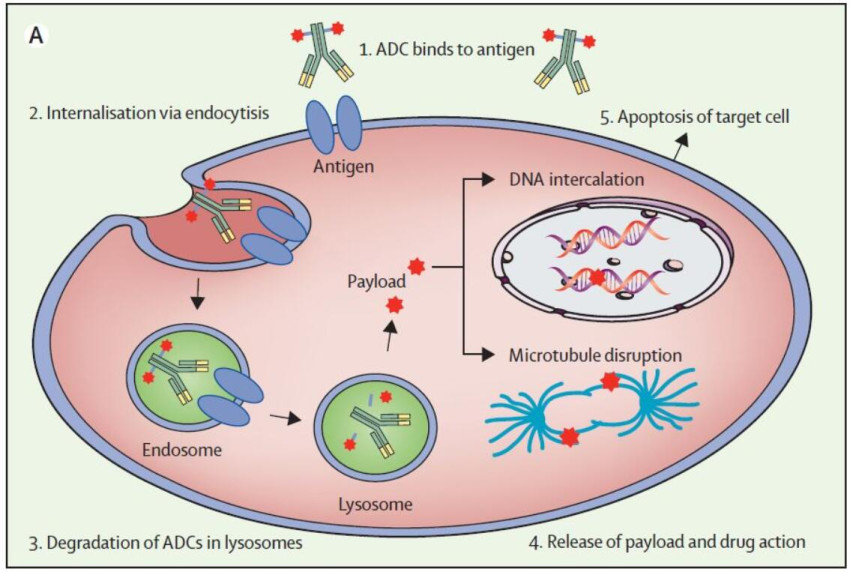
Antibody-drug Conjugate (ADC) is a new type of anti-tumor treatment strategy, which combines highly targeted monoclonal antibody (mAb) with a small molecule drug (payload) with strong cytotoxicity. A degradable or non-degradable linker (linker) connected to form a composite molecule. The principle of ADC is to use the high affinity and specificity of antibodies to precisely deliver drugs to overexpressed or specifically expressed antigens on the surface of tumor cells, and then release the drugs through endocytosis or hydrolysis of linkers to kill cells function, thereby achieving selective killing of tumor cells while reducing damage to normal tissues.
At present, ADC is mainly used in the treatment of hematological tumors and some solid tumors, and has made major breakthroughs in some indications. However, we have also noticed that in recent years, ADC has faced some challenges while developing rapidly, mainly including the following aspects:
1. Target selection
An ideal target should be highly specifically expressed on the surface of tumor cells, with low or no expression on normal cells. However, at present, most ADC targets also have a certain degree of normal tissue expression, which limits the maximum tolerated dose of the drug and reduces the efficacy. For example, CD30, the target of Brentuximab vedotin, is also expressed in some normal tissues, leading to side effects of peripheral neuropathy. Finding highly specific tumor targets is one of the current research hotspots in the development of ADCs.
2. Tumor heterogeneity
Tumor cells in the same type of tumor are highly heterogeneous, and different cells have different dependencies on drug targets. This may result in some tumor cells being insensitive or resistant to drugs. Ado-Trastuzumab has a significant effect in treating HER2-positive breast cancer, but HER2 weakly positive or negative tumors are not sensitive to it, and may grow into a dominant cell line after treatment. Strategies to address this issue include developing bispecific antibodies with multiple epitopes or improving the bystander effect of ADCs, etc.
3. Selection of linkers
Linker instability can lead to drug release outside the intended site, increasing toxic side effects. Gemtuzumab ozogamicin, which was first marketed in ADC, was caused by the instability of the linker, which caused severe toxicity of locked-in syndrome. The ideal linker should be highly stable in blood and can be rapidly cleaved in tumor cells to effectively release toxin molecules to the tumor site for killing. One of the highlights of Enhertu is that its new linker design significantly improves its blood stability and expands the therapeutic window.
4. Drug resistance of tumor cells
The generation of ADC drug resistance has multiple mechanisms, including: 1) tumor cells can evade drug killing by reducing or completely losing the target antigen targeted by ADC; 3) Tumor cells can activate drug resistance-related pathways such as ABC transporters to excrete cytotoxic drugs in ADC; 4) Tumor cells can activate other bypass signaling pathways to circumvent the main pathway of ADC inhibition; 5) Some tumor cells can reduce the release of toxins by changing the cleavage response of lysosomes to linkers; 6) Tumor-associated macrophages in the microenvironment can phagocytize ADC and reduce drug concentration.
5. The effect of tumor microenvironment
The tumor microenvironment such as tumor-associated macrophages also affects the delivery and efficacy of ADCs. Previous studies have found that tumor-associated macrophages can phagocytize antibodies through FCγ receptors and reduce the efficacy of ADCs. Understanding the influence of the microenvironment on drug efficacy and adopting corresponding strategies are also current research hotspots.
At present, ADC treatment of tumors has gradually become one of the important means of precision medicine for tumors. At present, 15 ADC drugs have been approved for marketing in the world, and are used in the treatment of lymphoma, leukemia and various solid tumors. The representative drug Enhertu has shown significant improvement in survival and remission rate in a number of clinical trials. With the continuous optimization of technology, the efficacy and safety of the new generation of ADCs are expected to be further improved. At the same time, factors such as tumor heterogeneity and drug resistance have also raised new problems for the development of ADCs.
The future development trend in the ADC field is to develop antibodies with more diverse targets and higher specificity, design new linkers to improve stability, and use ADCs in combination with other drugs to improve efficacy and overcome drug resistance. We have reason to believe that as more and more products are approved for marketing, ADC is expected to become the main choice for the treatment of some blood tumors and solid tumors.
As a leading CDMO for PEG derivatives and pharmaceutical intermediates supplier, Huateng Pharma can meet customers’ needs from lab research to pilot plant to large sacle commercial production. We are dedicated to being your most reliable partner to provide chemical synthesis and high-quality PEG linkers for ADC drugs.



