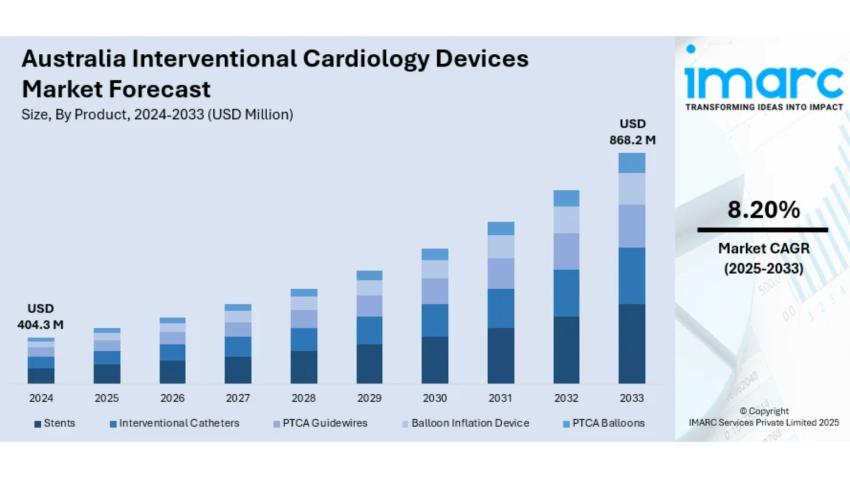
The latest report by IMARC Group, titled "Australia Interventional Cardiology Devices Market: Industry Trends, Share, Size, Growth, Opportunity and Forecast 2025-2033," is offering a comprehensive analysis of the Australia interventional cardiology devices market growth. The market size reached USD 404.3 Million in 2024. Looking forward, IMARC Group expects the market to reach USD 868.2 Million by 2033, exhibiting a growth rate (CAGR) of 8.20% during 2025-2033.
Base Year: 2024
Forecast Years: 2025-2033
Historical Years: 2019-2024
Market Size in 2024: USD 404.3 Million
Market Forecast in 2033: USD 868.2 Million
Market Growth Rate 2025-2033: 8.20%
Australia Interventional Cardiology Devices Market Overview
The Australia interventional cardiology devices market is growing steadily because there are more older people and more cases of heart disease. More hospitals are using less invasive procedures, which is making them want newer technologies. This helps hospitals give better care and make patients recover faster. Using AI in cardiac monitoring is helping doctors detect heart rhythm problems more accurately and also making their work easier. Drug-eluting stents are becoming more popular because they are effective in stopping the reblockage of arteries.
Australia Interventional Cardiology Devices Market Trends
• Using smart devices powered by AI in cardiac monitoring, like Medtronic's Reveal LINQ with its AccuRhythm system, helps make diagnoses more accurate.
• More doctors are using small, less invasive tools such as drug-coated stents that dissolve over time, along with various types of catheters for procedures.
• The range of products available now includes stents, PTCA guidewires, balloon inflation tools, and different kinds of balloons.
• Patients and doctors are increasingly choosing ambulatory surgical centers instead of hospitals for certain procedures.
• The market is growing in regions including the Australian Capital Territory, New South Wales, Victoria, Tasmania, Queensland, the Northern Territory, South Australia, and Western Australia.
Australia Interventional Cardiology Devices Growth Drivers
As the population gets older and people develop more heart-related risks because of their lifestyle choices, there is a growing number of heart problems and a higher need for medical procedures to treat them. There are also new improvements in medical technology and the design of devices, including the use of artificial intelligence. The healthcare system is expanding, making it easier for people to get heart care. Plus, getting approval from regulators is helping new products enter the market and encouraging innovation.
Australia Interventional Cardiology Devices Market Segmentation
Product Insights:
• Stents
o Drug Eluting Stents
o Bare Metal Stents
o Bio-Absorbable Stents
• Interventional Catheters
o IVUS Catheters
o Guiding Catheters
o Angiography Catheters
• PTCA Guidewires
• Balloon Inflation Device
• PTCA Balloons
o Cutting Balloons
o Scoring Balloons
o Drug Eluting Balloons
o Normal Balloons
End User Insights:
• Hospitals
• Ambulatory Surgical Centers
• Others
Regional Insights:
• Australia Capital Territory & New South Wales
• Victoria & Tasmania
• Queensland
• Northern Territory & Southern Australia
• Western Australia
Recent Developments of Australia Interventional Cardiology Devices Market
• Medtronic's AI-driven Reveal LINQ cardiac monitors were upgraded in May 2024 to mitigate false positives and improve arrhythmia detection.
• SMT's Supraflex Cruz drug-eluting stent received TGA approval in September 2024, with enhanced flexibility and drug delivery capabilities.
Competitive Landscape:
The market research report has also provided a comprehensive analysis of the competitive landscape. Competitive analysis such as market structure, key player positioning, top winning strategies, competitive dashboard, and company evaluation quadrant has been covered in the report. Also, detailed profiles of all major companies have been provided.
Australia Interventional Cardiology Devices Market News:
• September 2024: SMT's Supraflex Cruz drug-eluting stent received TGA approval in Australia, enhancing treatment options for coronary artery disease. This regulatory milestone strengthened the interventional cardiology devices market by introducing a clinically proven, flexible stent, supporting advanced patient care and driving market expansion.
• May 2024: Medtronic enhanced its Reveal LINQ cardiac monitors with AI-powered AccuRhythm algorithms in Australia, reducing false alerts for AFib and pause episodes. This upgrade improved diagnostic accuracy and clinical efficiency, strengthening Australia's interventional cardiology devices market with advanced, AI-driven monitoring solutions.
Note: If you require specific information not covered in this report, IMARC Group offers customized research solutions tailored to your business needs.
About Us
IMARC Group is a leading market research company providing management strategy and market research worldwide. We partner with clients across sectors and regions to identify high-value opportunities, address critical challenges, and transform businesses through comprehensive market intelligence and actionable insights.
Contact Us
134 N 4th St. Brooklyn, NY 11249, USA
Email: sales@imarcgroup.com
Tel No: (D) +91 120 433 0800
United States: +1-631-791-1145




